The number of AI-enabled medical devices granted FDA marketing authorization for radiology surged past the 1k mark in the latest update from the agency. The numbers show that radiology’s share of authorizations remains stable at just over three-quarters of total approvals.
The FDA regularly releases the list in what’s become a closely watched barometer of both total approvals as well as which medical specialties are most active in AI.
- Radiology has historically garnered the lion’s share of approvals – perhaps no surprise given the discipline’s early adoption of both digital image management and AI – with the first authorization granted in 1998 (for ImageChecker mammography CAD from R2 Technology/Hologic).
The new list tracks authorizations through the end of September 2025, and indicates the agency has…
- Authorized 1,356 AI-enabled devices since it started tracking, up 8.5% since its last report.
- Approved 1,039 AI-enabled radiology devices, with imaging accounting for 77% of total medical authorizations since 1998.
- Radiology secured 75% of total authorizations from June to September (83/110), compared to 78% from January to May 2025, 73% for all of 2024, and 80% for 2023.
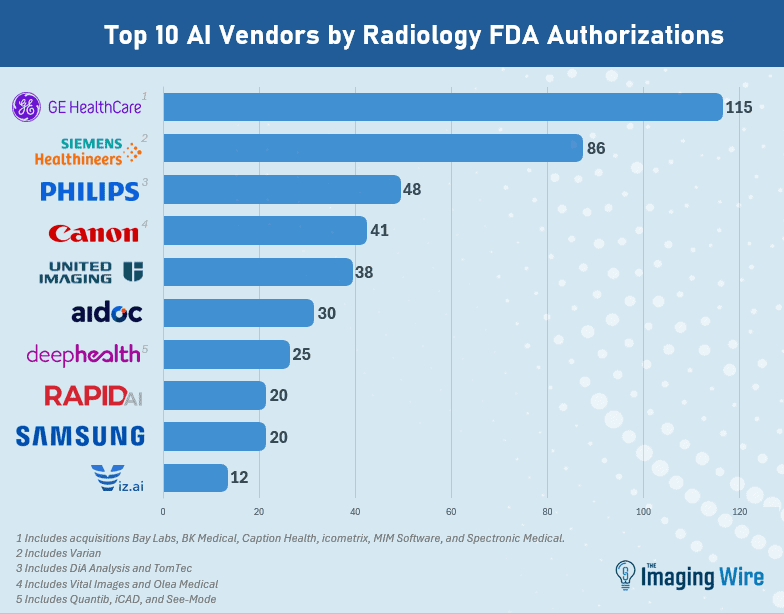
- GE HealthCare retains the top spot as the company with the most radiology AI authorizations, at 115 (including recent acquisitions Bay Labs, BK Medical, Caption Health, MIM Software, icometrix, and Spectronic Medical).
- Next is Siemens Healthineers at 86 (including Varian), then Philips at 48 (including DiA Analysis and TomTec), Canon at 41 (including Vital Images and Olea), United Imaging at 38, and Aidoc at 30.
As always, it’s worth noting that the FDA’s list includes not only standalone software applications, but also imaging equipment that might have AI applications embedded into it, such as a mobile X-ray system with AI algorithms for detecting emergent conditions.
- Also, the agency noted that it is exploring ways to identify and tag AI-based devices that use foundation models and large language models. The FDA has yet to approve an LLM-based medical device.
The Takeaway
The new numbers indicate that radiology’s dominance of medical AI continues. But they also show that the FDA has returned to a regular twice-yearly cadence of updating its list of AI-enabled medical devices after a break of nearly a year – news that’s welcome to AI developers.